Marching Forward with Food Analysis
The analytical aspect of the food industry has changed significantly over the last decade – mainly because of advances in technology. From pesticide residues to metabolomic approaches, the field is now more exciting than ever.
sponsored by Thermo Fisher Scientific
I’ve been with Fera (in its various guises) for 12 years now. I joined for what I thought would be a single year of good experience after finishing my BSc in Biology at the University of York. But I never left – I was hooked. After two years, I’d made it into a research team, and was lucky enough to work with food analysis research scientist Richard Fussell (who recently joined the team at Thermo Fisher Scientific).
My time at Fera so far has given me an excellent and broad view of the field. In the early years I was involved in the application of emerging mass spectrometry techniques for the multi-residue determination of pesticides and veterinary medicines in food. I also looked at the fate and behavior of pesticides, veterinary medicines and pharmaceuticals in the environment. Twelve years isn’t such a long time in terms of analytical chemistry, but during that time, mass spectrometry has advanced at a furious pace. In more recent years, high-resolution (HR)-MS systems started to appear in our laboratories and, as luck would have it, I began working on fate and behavior projects that could benefit from HR-MS. Now, I find myself working almost entirely in the world of HR-MS, including TOF, QTOF, and Orbitrap instruments. I lead metabolomics at Fera in Adrian Charlton’s section and essentially bring LC- and GC-MS expertise alongside NMR-spectroscopy for biochemical profiling. We aim to exploit fully the complementary nature of the three approaches; NMR gives excellent reproducibility and you can quantify without analytical standards – but sensitivity can be an issue. LC- and GC-MS systems offer additional coverage, especially in terms of sensitivity. We’re currently involved in a EU project called ABSTRESS (www.abstress.com), which is looking at dual stress in legumes. There have been many studies addressing resistance mechanisms in drought-stressed and disease-stressed plants individually, but with dual stress the biochemistry in the plant can be different. We’re using metabolomics and transcriptomics to identify the hub genes that are crucial for dual stress resistance, working with 12 national and international partners in the EU, including a state-of-the-art plant phenotyping platform at INRA (the French National Institute for Agricultural Research). It’s exciting work.
The IC-MS story
In addition to cutting-edge research that addresses global food challenges, one of the big goals at Fera is to improve analytical throughput and efficiency. Back in 2008, Richard Fussell and I embarked on a project to try and combine single residue methods for polar ionic pesticides (PIPs), such as glyphosate, chlormequat, mepiquat, ethephon – the project continues today with Stuart Adams, Senior analytical chemist at Fera. I recently gave a lecture that summarizes the project and outcomes at the 1st International Symposium on Recent Developments in Pesticide Analysis called “IC-MS Multi-residue pesticide methods, fantasy or reality?”
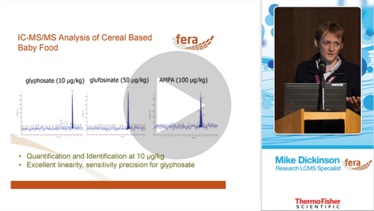
In pesticide analysis, multi-residue methods are king; they enable higher throughput and increase laboratory efficiency. Over the years, analytical chemists have done a fantastic job of shoehorning a high number of pesticides into such multi-residue methods. Unfortunately, several stubborn stragglers refuse to be constrained – either they don’t quite fit with the chromatography chemistry or the MS analysis is not ideal. Not only are PIPs difficult to separate, they need to be quantified and identified at low concentrations (for example, 0.1 µg/L in drinking water). And the truth is that even the single residue methods we do have are not particularly successful for certain compound-matrix combinations; for example, glyphosate at low µg/kg concentrations in maltodextrin products.
In fact, glyphosate was a big reason why we decided to investigate the potential of ion chromatography (IC) in pesticide residue analysis. Glyphosate is the most used pesticide throughout the world – people have been splashing it over their gardens for year (as an aside, renewed interest in its toxicity has been hitting the headlines recently). Glyphosate is also one of the most difficult compounds to analyze. Fortunately, it is amphoteric (can exist in different ionic forms) and that triggered a question: why not use IC?
One hurdle was the perceived difficulty in hyphenating IC with MS; MS systems don’t get along with salt mobile phases (potassium hydroxide, in our case). The introduction of robust and reliable membrane electrolytic suppressors to convert potassium hydroxide to water on exit from the column was a game changer. Nevertheless, we still had our concerns, and kicked off the project using a relatively old MS instrument, primarily to protect the expensive MS systems – against damage should the suppressor fail. Despite the antiquity of the MS system, we started getting really promising results for retention of glyphosate and glufosinate (another systemic herbicide) using our Dionex ICS-3000. More important were the findings of transformation products – AMPA (from glyphosate) and MPPA and N-Acetyl glufosinate (from glufosinate) – all of which are in the residue definitions for either food or water, or both. We had to load 5-ml samples into the system using an inline concentrator pre column; you can probably imagine the state of the MS source after a series of injections, given that we were not only concentrating our compounds of interest but also matrix co-exatractives. Maltodextrin products proved especially challenging.
The pesticide lab of tomorrow
In terms of IC-MS for pesticide analysis, we’ve learnt a number of tricks along the way (for example, writing a script that shut down the pump in the case of a consistent offscale detector response, which can be indicative of a suppressor failure emergency) – and we benefited from the work of Anastassiades et al., in particular the QuPPe method (1). Indeed, Stuart has continued to develop the method, extending it to other pesticides of interest as well as some other stragglers that don’t fit particularly well into multi-residue methods. Notably, we’ve also moved onto much more advanced triple quadrupole MS systems and we now inject only 100 µl samples because of advances in sensitivity. The use of internal standards was also another big step forward, and I suspect another area where we can expect to see further benefits.
Fera has an upcoming collaborative research project with Thermo Fisher Scientific that will employ a state-of-the-art system – a Dionex ICS-5000 coupled to a TSQ Quantiva triple quadrupole mass spectrometer. It’s a combination that promises to give us the highest possible levels of sensitivity, and also offers the potential to enter into the world of 2D-LC. The ability to use conventional reversed-phase chromatography on the first dimension column and IC on the other would certainly help separate some of the trickier compounds from the matrix. We could also run two columns in IC mode, using the second column as a concentrator before MS analysis. Stay tuned for further developments!
Looking to the future makes me think about how quickly things have changed. Walking through the laboratories at Fera is very different today than when I started just 12 years ago; there is less wet chemistry with fewer “hands on” analysts, and the instrumentation is much more sophisticated, which has increased efficiency immensely. Technicians no longer need to spend hours concentrating samples because the equipment provides the sensitivity we require, especially with the revolution in triple quadrupole instruments.
We now have more than 35 MS systems that deal with routine analyses (the majority are LC-triple quads, with about five HR-MS instruments). And though the triple quadrupole instruments are very sensitive, reliable and ideal for quantification and targeted methods, it wouldn’t surprise me if hybrid quadrupole high-resolution instruments start to take over at some point in the future. Instruments like the Q Exactive offer good sensitivity for targeted analysis alongside the potential for full-scan, high-resolution data acquisition.
Looking further into the future, there will no doubt be a continuation in the development of hardware, but software is likely to result in breakthroughs with the biggest impact in my opinion. Indeed, software companies and developers must focus on providing user-friendly, efficient, fully capable solutions. I also expect to see scaling down of instruments to drive towards portable equipment that can be operated by non-experts. We’ve already seen the beginnings of a revolution in NMR benchtop devices like the Picospin, so why not with MS instruments? The core technology may not be there quite yet, but it will come. Miniaturized Hybrid Quadrupole-Orbitrap anyone? I’ll
take two.
- M Anastassiades et al., “Quick method for the analysis of numerous highly polar pesticides in foods of plant origin via LC-MS/MS involving simultaneous extraction with methanol (QuPPe Method, Version 8.1 www.eurl-pesticides.eu/library/docs/srm/meth_QuPPe.pdf, 2015)
Mike Dickinson, Research LCMS Specialist, Fera Science Ltd., Sand Hutton, UK.