Solving Pharmaceutical QC Pain Points: Advanced Lab Balances for Accuracy and Compliance
Solutions for Precision, Compliance, and Efficiency in Pharma QC
In pharmaceutical quality control, precision and compliance are crucial. Lab balances, such as the Cubis® II series, play a vital role in addressing industry challenges. These balances ensure data integrity, secure user access, and reliable data transfer, meeting stringent regulatory standards like 21 CFR Part 11 and EU Annex 11. Explore the attached documents to discover how Sartorius solutions can transform your QC processes, ensuring reliable and efficient pharmaceutical production.
In pharmaceutical quality control, precision and compliance are crucial. Lab balances, such as the Cubis® II series, play a vital role in addressing industry challenges. These balances ensure data integrity, secure user access, and reliable data transfer, meeting stringent regulatory standards like 21 CFR Part 11 and EU Annex 11. Explore the attached documents to discover how Sartorius solutions can transform your QC processes, ensuring reliable and efficient pharmaceutical production.
Unique Features:
Ionization Technology: Complete elimination of static interferences to enhance performance.
Low Minimum Sample Weight: Reduces sample waste and improves sustainability.
Monolithic Weighing System: Provides more accurate and repeatable measurements.
Calibration Certificates from Accredited Providers

This white paper explains the basic concepts of accredited calibration and the advantages of certificates from accredited providers, focusing on ISO/IEC 17025 accreditation for calibration laboratories.
DownloadProtecting Data Integrity - Evaluating Instruments in the Lab

Ensure compliance with data integrity regulations by evaluating your lab instruments and procedures using this comprehensive checklist.
DownloadMinimum Sample Weights According to USP, OIML R 76, and EURAMET cg-18

Understand the concepts of specifying minimum sample weights to avoid exceeding the specified relative measurement uncertainty.
DownloadUse of Laboratory Balances in the Pharmaceutical Industry
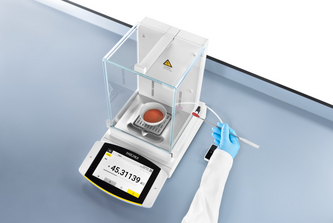
This white paper presents testing requirements for lab balances as described in pharmacopoeia chapters, along with Sartorius Service certificates.
DownloadCubis® II Pharma Compliant by Design
Discover how the MCA high-end 7” display and QApp pharma package meet pharmaceutical industry requirements for a compliant lab balance system.
DownloadJapanese Pharmacopeia Chapter 9.62 Measuring Instruments, Appliances
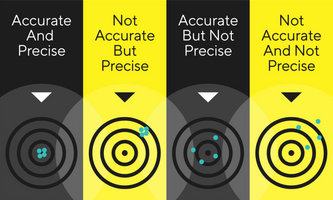
Learn about the recent updates to the Japanese Pharmacopeia, including new chapters on balances and weighing in the "General Information" section.
DownloadSample Prep for Quality Control
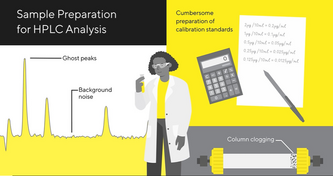
Quality control is crucial in the biopharmaceutical and medical device industry. Sartorius offers analytical sample preparation solutions for ultra-trace level detection in HPLC, LC-MS, IC, and ICP-MS, supporting manufacturers in meeting the increasing QC requirements of global regulatory agencies for safe, potent, and pure products.
Download