In 2013, Barack Obama announced the US BRAIN Initiative, a 10-year research endeavor to understand how the brain works. For Bhavya Sharma, Assistant Professor of Chemistry at the University of Tennessee Knoxville, USA, this was a springboard for her own brain imaging research.
“I had learned that, in general, brain imaging techniques provide high spatial resolution but little to no chemical information. Perhaps more importantly for the owner of the brain, techniques that provide chemical information often require holes to be drilled in the skull or portions of the skull to be removed to access brain tissue,” she says. Sharma saw Raman spectroscopy as a way to get the information researchers need without physically penetrating the skull. And she knew it could have a big impact. “I believed that if I could make this work, we would be able to develop Raman-based methods for neurological disease diagnosis, particularly for diseases that are difficult to diagnose in the early stages, such as Parkinson’s disease,” she says. In an initial study, Sharma and her team used animal skulls filled with agarose gels to mimic brain tissue, combining surface-enhanced Raman spectroscopy (SERS) and spatially-offset Raman spectroscopy (SORS; combined name: SESORS) to look inside the skulls (1).
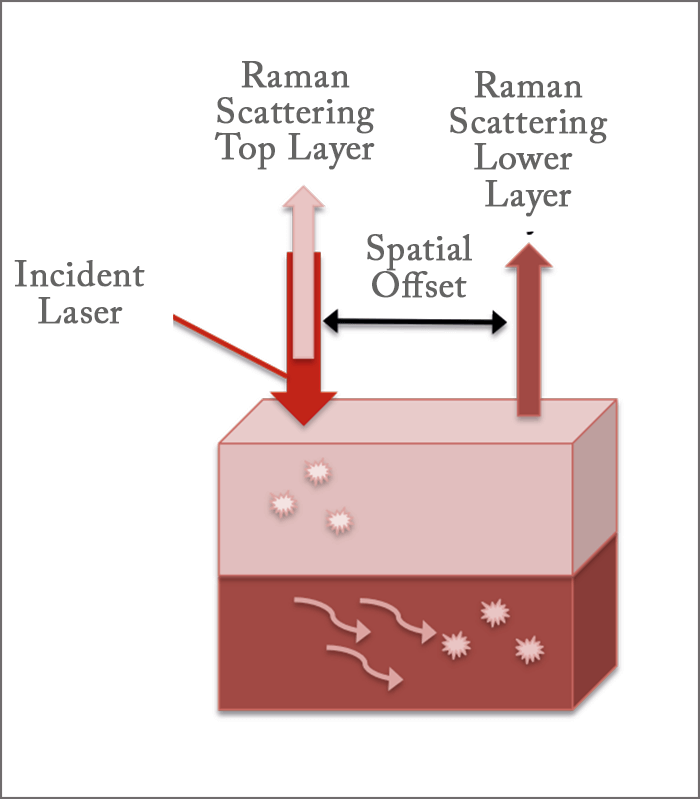
“SERS utilizes noble metal nanoparticles to enhance the inherently weak Raman scattered light through an electric field generated at the surface of the nanoparticles, allowing us to collect Raman spectra of analytes down to the nanomolar range),” she says. “In SORS, the laser is incident on the surface of a multilayer sample, with the Raman light scattered from the top layer traveling back along the same trajectory as the incident laser. Some of the photons travel into the lower layer and migrate laterally before being scattered out. The distance between the incident laser and where the light is scattered from the lower layer is the spatial offset.” By collecting the Raman scattered light at the spatially-offset location while going through the top layer, the team gained information about the lower layer (see Figure 1). They were able to measure the Raman spectra of three different neurotransmitters through skull bones across a concentration range of 100 micromolar (µM) to 500 millimolar (mM). But they’re keen to take it to the next level. “The measurements we performed were with skulls that are two mm thick, but in humans, the skull is 3-14 mm thick,” she says. “Our challenge now is to lower our limits of detection and increase our depth of penetration – but at this point we’re already within one mm of measuring the thinnest part of a human skull.” Sharma believes that SESORS holds great promise for use in non-invasive measurement of neurochemicals in the brain. “We are moving forward with new instrument designs, and have moved onto measuring skulls with greater thicknesses,” she says. “Our preliminary results are promising – so stay tuned… We expect to have a lot of new and exciting results to share in the future!”
References
- AS Moody et al., “Surface-enhanced spatially offset raman spectroscopy detection of neurochemicals through the skull”, Anal Chem, (2017). DOI: 10.1021/acs.analchem.7b00985




