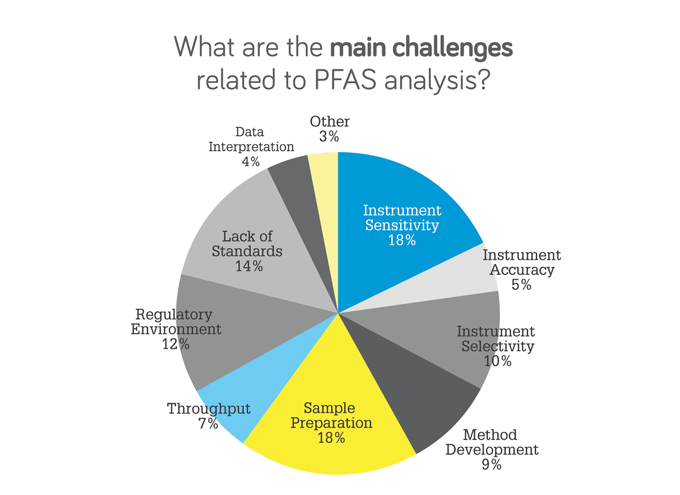
What are the main challenges related to PFAS analysis?
Powell: When I first started looking at PFAS, there were only two compounds PFOS and PFOA, but according to the most recent estimations, there could be over 13,000 different PFAS compounds in the environment. Extremely low detection levels and background issues can be a real challenge for labs. Multiple solvents and consumables can contain PFAS, so if you can’t get a blank, you’ll never get down to the very low detection levels. Regulatory challenges are also very important.
Chadha: Yes, the lack of standards is a key one. For years, we’ve only had one real provider of standards, Wellington Laboratories. It’s crucial that we have a second supplier through the validation process – and we at Agilent are trying to provide an alternative. We also have to make sure that our methods are fast and robust. In addition, we also need to make sure that, as we’re detecting more PFAS through our screening methods, we’re adding these into our targeted methods.
Marf il-Vega: Without standards, instrument sensitivity or sample preparation are irrelevant, and we’re stuck analyzing the same 40-or-so targets.
Waclaski: Agreed. I’ll also add that sample preparation can be quite tedious, with opportunities for human error. And that’s why customers are always interested in ways to automate sample preparation. Contamination is another big challenge because it can affect sensitivity; you have to be careful to use high-grade materials for your sample preparation.
Are current analytical methods for PFAS analysis sufficient?
Marf il-Vega: It is true that, in general, we know that for the thousands of chemicals out there, we do not have standardized methods for all sample types. However, I think that in some fields, such as environmental, food, and even clinical, work has been ongoing for decades. We have accumulated a lot of knowledge that can now be transferred to emerging areas where we are just starting to analyze PFAS.
Chadha: A d egree o f u ncertainty is expected because this is an ever-evolving area in terms of regulations, requirements, customer needs, and the needs from different industries. I think we’ll always be always catching up.
Waclaski: I would agree with Ruth and Marcus – and add that there’s just so many PFAS compounds out there. We’re looking at thousands of potential compounds, which makes it really difficult to quantify. I’m specifically working with a lot of non-targeted methods, which can look at these as a broad class of organofluorine compounds with a little bit of a different approach. But there are limitations with that too because you don’t know exactly which compounds you have.
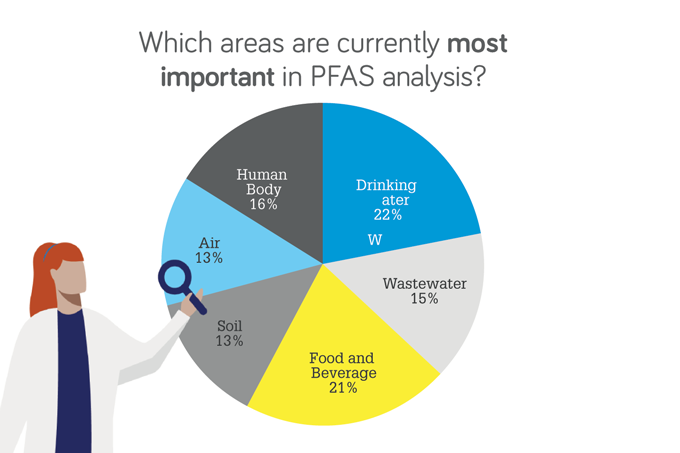
Which areas are currently most important in PFAS analysis?
Chadha: The sur vey results are broadly what I would have expected and likely reflect where the regulations appeared first. In the coming years, I think monitoring people’s exposure at the workplace will increase, as will air monitoring near high risk areas, such as incineration plants. For food, we’re going to see more testing of materials that come into contact with packaging.
Marf il-Vega: I would like to add that although drinking water is an important area for PFAS analysis, the PFAS must be coming from somewhere else – either in the products we use daily or industrial waste. So I think we’re going to see a greater focus on where the PFAS are created in the first place.
Powell: A greed. F inding t he s ources that are leading to drinking water is a big challenge and includes consumable products, wastewater, and more.
Waclaski: I am seeing an increase in consumer product testing. But the number of sources is growing, and includes food packaging, cosmetics, carpet fibers, clothing – you name it. We see companies trying to make alternate compounds that are certified PFAS free – this is a growing area and there’s a lot of testing involved there.
Day Powell is Environmental Application Specialist at Agilent; Marcus Chadha is EMEA Applications Specialist Manager, also at Agilent; Linx Waclaski is Product Manager for Ion Chromatography at Metrohm USA; and Ruth Mar f il- Vega is Senior Market Manager for Environmental at Shimadzu.
Dive deeper with Marcus Day, Linx, and Ruth as they discuss trends and challenges in PFAS analysis in this on-demand webinar: https://bit.ly/3SR1LBN
Marcus Chadha and Day Powell: How does Agilent achieve low detection limits for PFAS with its targeted methods? Our strategies include: sample enrichment – an offline SPE step that allows you to preconcentrate your sample and then inject it onto our most sensitive LC-MS instruments. It’s more labor intensive, but it does allow us to get down to the parts-per-quadrillion levels. In addition, online SPE LC-MS allows us to do very large injections, followed by an online SPE step. The configuration is more complicated, but, once set up, it’s less labor intensive than the offline SPE and the instrument handles the extraction for you. Lastly, we have LC-MS with direct injection. This is the simplest and easiest way to get down to really low levels and optimize blanks.
For these methodologies, we have solutions to help labs get their PFAS methods set up. We have our PFC-Free HPLC Conversion Kit for our instrumentation, which minimizes PFAS from the system. We also have our PFC-Free Columns and Supplies, which includes consumables and helps eliminate PFAS from the actual prep of your samples. Finally, in addition to Application Services and Support, our PFAS MRM Database contains hundreds of different PFAS compounds optimized across our instrumentation.
Feasibility studies to remove PFAS
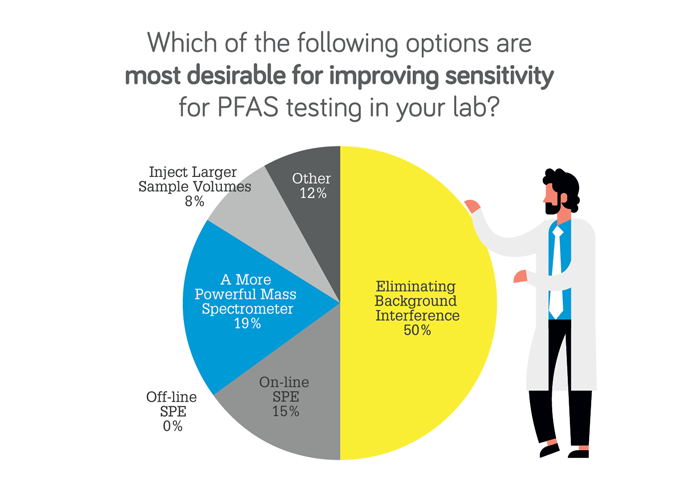
Ruth Marfil-Vega: Sensitivity is often cited as one of the biggest challenges in PFAS analysis – often people will say you need a triple quadrupole LC-MS instrument with the highest sensitivity possible. This is driven by regulatory requirements enforcing limits of detection at nanograms-per-liter concentrations. But there are other parameters besides the sensitivity of the instrument that are important to keep in mind. For example, when doing feasibility studies for the removal of PFAS, samples are normally spiked at micrograms-per-liter or even milligrams-per-liter levels. Here, a triple quad is not needed. Instead, a single quad can work well to quantify the individual PFAS present in the sample.
Shimadzu has used a single quadrupole LCMS-2050 to target 28 PFAS. We used the standard DUIS ionization source, simultaneous acquisition in scan, selected ion monitoring, and an injection volume of 1 uL. We were able to calibrate the instrument between 0.5 and 200 ng/mL, our limits of quantitation were in the ng-per-mL range, with less than 12 percent relative standard deviation and an accuracy between 80-120 percent. This work demonstrates that you don’t always need a triple quad instrument for PFAS analysis.
Combustion ion chromatography for non-targeted PFAS analysis
Linx Waclaski: Non-targeted analysis of PFAS encompasses a broad class of compounds, which include those containing organic fluorine. Non-targeted analysis is thought to be a better risk assessment tool for measuring the true impact of fluorine on the environment. An emerging technique uses a specialized sample preparation, followed by combustion ion chromatography.
There are several approaches for non-targeted analysis of organic fluorine combustion IC. The hottest method today involves measuring absorbable organic fluorine (AOF) by first capturing PFAS compounds from 100 mL of an aqueous sample using granular-activated carbon, then washing off inorganic fluoride with nitrate solution, before combusting the carbon to measure AOF via combustion IC, with an approximate detection limit of 2–5 ppb.
The excitement around this method is reflected in the release of DIN 38409- 59 and the development of the Draft EPA Method 1621, both of which incorporate AOF.