Introduction
Nanotechnology is an emerging and rapidly growing field whose dynamics and prospects pose many great challenges to scientists and engineers. Nanoparticles are being used in many materials and products, including coatings (on plastic, glass and clothing), sunscreen, antimicrobial bandages and clothing, MRI contrast agents, biomedical elemental tags and fuel additives, only to name few. However, rapid, simultaneous characterization of their elemental composition, number of particles, size, and size distribution is challenging. For inorganic nanoparticles, the technique best suited to provide the above-mentioned characteristics is inductively coupled plasma mass spectrometry (ICP-MS) operated in so-called single particle mode. Analyzing single nanoparticles with ICP-MS requires a different approach than measuring dissolved elements. This work describes the theory behind single-particle ICP-MS measurements, drawing comparisons and differences with analyzing dissolved elements.

Understanding Single Particle ICP-MS Analysis
Effectively detecting and measuring single, individual nanoparticles with ICP-MS requires operating the instrumentation in a different manner than when analyzing dissolved samples. Figure 1 shows traces from both dissolved and single nanoparticle analyses. In Figure 1a, a steady state signal results from measuring dissolved elements; the output when detecting single particles is quite different, as illustrated for 60 nm silver particles in Figure 1b. Each spike in Figure 1b represents a particle. The differences in the way these data are acquired are the key to understanding single particle analysis. The easiest way to gain this understanding is to review and compare the processes involved when both dissolved elements and particles are measured.
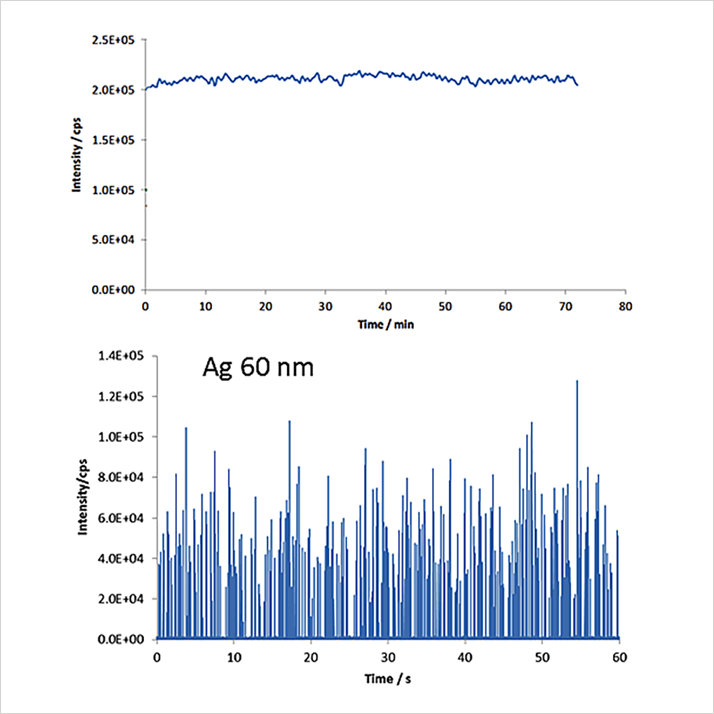 Figure 1. a) A continuous signal from measuring a dissolved analyte; b) A signal from measuring 60 nm silver nanoparticles.
Figure 1. a) A continuous signal from measuring a dissolved analyte; b) A signal from measuring 60 nm silver nanoparticles.Dissolved Analyses with ICP-MS
When dissolved elements are measured, aerosols enter the plasma, where the droplets are desolvated and ionized. The resulting ions enter the quadrupole to be sorted by their mass-to-charge ratios (m/z). The quadrupole spends a certain amount of time at each m/z before moving to the next m/z; the time spent analyzing each m/z is called "dwell time". After each dwell time measurement, a certain amount of time is spent for the electronics to stabilize before the next measurement is performed. This stabilization time is called “settling time”, i.e. overhead and processing time. When analyzing dissolved elements, the resulting signal is essentially a steady-state signal, as shown in Figure 2a. However, considering the dwell and settling times, a significant amount of the signal is not measured due to the settling time of the electronics, a critical aspect when analyzing nanoparticles (Figure 2b).
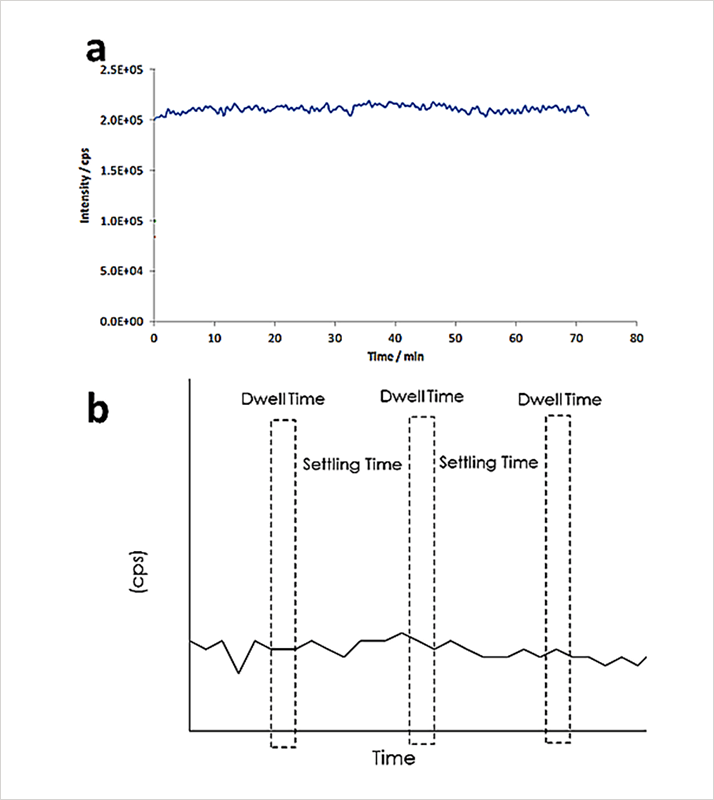 Figure 2. a) A continuous signal from measuring a dissolved element; b) A continuous signal, with the dwell and settling times overlaid – data is only collected during the dwell time.
Figure 2. a) A continuous signal from measuring a dissolved element; b) A continuous signal, with the dwell and settling times overlaid – data is only collected during the dwell time.




