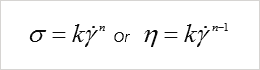Abstract
A material’s rheological properties not only influences its visual and textural perception, but also affects its processing capabilities. For instance, compared to Newtonian materials, shear-thinning materials are more susceptible to applied stress.
Introduction
While most suspensions and polymer structured materials are shear thinning, some materials can also show shear thickening behavior where viscosity increases with increasing shear rate or shear stress. This phenomenon is also often referred to as dilatancy, and although this refers to a specific mechanism for shear thickening the terms are often used interchangeably. In most cases, shear thickening occurs over a decade of shear rates and there can be a region of shear thinning at lower and higher shear rates.
Usually dispersions or particulate suspensions with high concentration of solid particles, pastes, associative polymers such as HASE, HEUR polymers etc. exhibit shear thickening. Materials exhibiting shear thickening are much less common in industrial applications than materials exhibiting shear thinning, however, where encountered shear thickening materials can lead to severe processing problems. Materials which undergo micro-structural or orientation changes on application of shear, that lead to increased resistance to flow, will tend to show shear thickening.
For suspensions this generally occurs in materials that show shear thinning at lower shear rates and shear stresses. At a critical shear stress or shear rate the organized flow regime responsible for shear thinning, is disrupted and so called ‘hydro-cluster’ formation or ‘jamming’ can occur. This gives a transient solidlike response and an increase in the observed viscosity. Shear thickening can also occur in polymers, in particular amphiphilic polymers, which at high shear rates may open-up and stretch, exposing parts of the chain capable of forming transient intermolecular associations.

η is the power law index
σ is the shear stress
ý is the shear rate With n greater than 1 for shear thickening fluids. It should be noted that an upturn in viscosity at high shear rates can occur through other phenomenon such as fluid turbulence. This effect, however, tends to occur with lower viscosity fluids and can be predicted from Reynolds number calculations.