Molecular imaging based on mass spectrometry provides a broad scope of analytical, molecular and local information that can be employed for patient phenotyping. Within M4I – the Maastricht MultiModal Molecular Imaging institute – we develop innovative technologies to generate and discover more detailed knowledge that enables surgeons to perform tissue typing ‘on-the-fly’.

Coming from a technological environment (FOM-AMOLF), I felt that we needed to translate our new imaging technologies into the clinic – becoming one of two directors at M4I enabled me to do that. The possibility to collaborate closely with Steven Olde-Damink (a surgeon), Peter Peters (a nanobiologist) and Clemens van Blitterswijk (regenerative medicine) was an enormous motivator for me and it also essentially allows me to take my research to a higher level – in the pursuit of personalized medicine.
Personalized medicine requires different molecular datasets to be generated in a concise manner. Imaging mass spectrometry is a unique discovery method and I would say the main enabling high-throughput technology for this purpose. Personalized diagnosis relies on the quick and complete characterization of, for example, patient biopsies. Once we gain sufficient information, it can be directly employed to ‘train’ smart surgical devices, such as the iKnife.
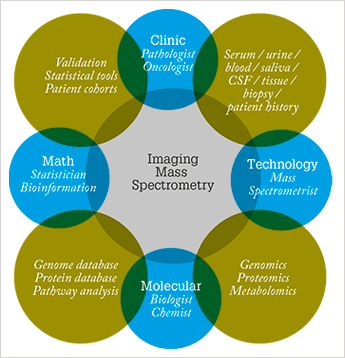

Maastricht University and the regional government have invested to strengthen the knowledge infrastructure for life sciences and health so that we can lead the European molecular imaging scene. Indeed, M4I offers a unique combination of enabling technologies for personalized medicine at the molecular scale, the cellular scale, the tissue scale and, most importantly, at the systems level scale. In particular, we are developing new technologies that push the boundaries of MS imaging. We strive to enhance throughput, speed, spatial resolution, sensitivity and molecular resolution with a combination of fundamental, instrumental and applied research. We cover the whole story from nanoscopy to patient diagnosis within one institute, and everything must be embedded within clinical practice. And that isn’t easy; the biggest hurdle was a logistical one – how could we house dozens of researchers and instruments in an existing infrastructure and bring everything up to the state of the art?
The answer was getting the right people. The new molecular imaging teams within M4I have the right expertise and capabilities to conduct truly translational research. The team is made up of a broad range of analytically-driven scientists, including (but not limited to) physicists, (bio-)chemists, material scientists, bioinformaticians, pathologists and clinicians. As a young university, Maastricht offers a stimulating environment. Facilitating and embracing multidisciplinary teams is a prerequisite for success in a day and age where the problems we tackle have become too complex for a single discipline. I think the unique element in our new endeavor is that different scientific cultures are able to open up and collaborate without hesitation. Finding surgeons who are willing to ask researchers how they can best optimize surgical protocols to improve personal molecular diagnosis is a real eye-opener. And it is indicative of the innovative attitude found at the Maastrict University Medical Center (MUMC).
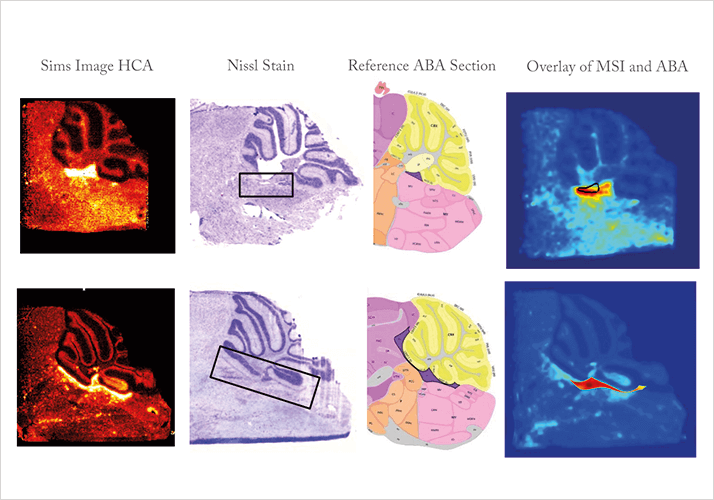
Success will come from a shared goal. The surgeons and the rest of the team at M4I know that better molecular diagnosis will result in more targeted treatment, which will in turn improve the patient’s prognosis and reduce the use of less effective therapies. For example, precise surgical margin determination during surgery reduces the chance of cancer recurrence, which has a clear impact on the quality of life for patients in the clinic. One thing is clear. The institute’s output in three years must feed directly into the clinic and contribute to improve health care. That is how we will make personalized medicine a reality.




