Since the advent of the industrial age, we’ve been in contact with synthetic chemicals – in our food, in the water, the dust, and the air. Analytical science has had to respond to the evolving challenges in assessing the routes and magnitudes of exposure to persistent organic pollutants (POPs) in different population segments, and I believe it is now on the verge of exquisite sensitivity.
Prediction or precision?
You can assess human POP exposure in two ways – prediction or measurement. Prediction relies on measuring POPs in the various media we come into contact with and on questionnaire data, which indicate an individual’s exposure to those media. Mathematical models are needed to derive a predicted internal dose in this conventional human health risk assessment approach. By contrast, biomonitoring involves direct measurement of POPs in tissue samples taken from an individual, directly quantifying the internal dose, and thus removing the need for assumptions. Operation Ranch Hand (the codename given to the defoliation program sprayers during the Vietnam War) resulted in dioxin-exposure to these veterans (specifically, 2,3,7,8-tetrachlorodibenzo-p-dioxin). The measurement of dioxin in these veterans nicely proves the point; predicted dioxin exposure level per veteran had no significant correlation with the actual serum dioxin measurements provided by biomonitoring. The moral of the story: don’t predict – measure! Many POPs, including dioxins, are lipophilic, and some dioxins can have half-lives in human tissues of ten years or more, enabling a direct measurement of exposure years after the event, using biopsy analysis. For lipophilic POPs, adipose tissue seems the obvious sample choice. But it may not be the best one. Back in the seventies, the US Centers for Disease Control (CDC) investigated the Times Beach incident, where a town in Missouri had become contaminated with dioxins after contaminated oil had been sprayed for dust control. I was one of the lead investigators at CDC.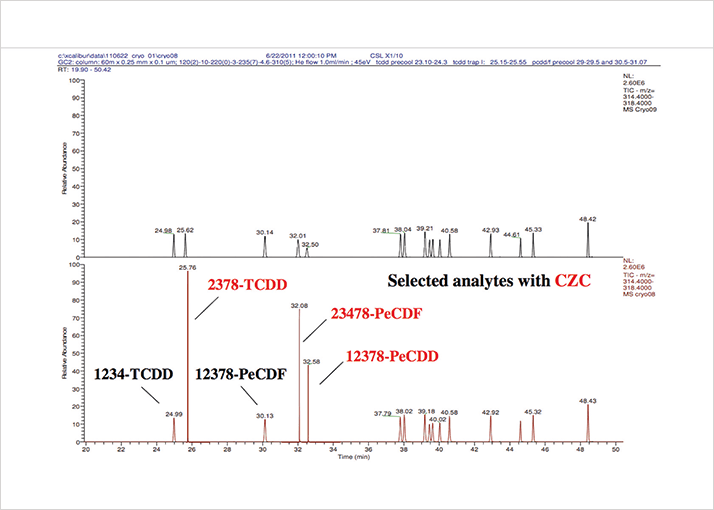
To measure exposure, we asked residents to submit to a surgical procedure that removed a fairly large 20 gram sample of subcutaneous fat from the abdominal area (such was the sensitivity of the technology available). The procedure was invasive – leaving what you might call a fairly large dimple – and, unsurprisingly, the participation rate was low. Soon afterwards, we looked into using serum instead and demonstrated a very tight correlation with the adipose measurements (1). Thankfully, dioxin biomonitoring no longer demands a pound of flesh...
However, although serum sampling is more tolerable for study participants, it presents analytical scientists with a sensitivity problem. Adipose tissue is ~95 percent lipid, and accumulates lipophilic POPs in the parts per trillion to the parts per billion range. But serum averages only about 0.6 percent lipid, which requires detection sensitivities of parts per quadrillion (ppq). Rigorous quality assurance is needed to eliminate background contamination at these levels, and we did everything we could to get reliably low backgrounds, banning smoking (before no-smoking policies were common) and stopping janitors cleaning the floors (phenolic products were disrupting analyses).
Attaining the required sensitivity, though difficult, enabled us to establish the ‘normal levels’ of POP exposure in the population; you must understand what is normal before you can know what is abnormal – and in industrialized societies, exposure to POPs is ubiquitous – we all have dioxins in our tissues. One ongoing initiative at the CDC assesses the exposure of the US population to >300 environmental chemicals; survey data get collated into the National Report on Human Exposure to Environmental Chemicals, which offers very valuable data about POP exposure, by sex, age and race/ethnicity, over time. Indeed, they’ve picked up some very interesting epidemiological points. For example; older people (over 60) typically have an internal dioxin dose three times that of people aged 20–39. And there are large differences in p,p-DDE (dichlorodiphenyldichloroethylene) levels between males and females in some ethnicities, but not in others. This information helps us to define ‘normal’ exposure for the US population.
Tools for the task
None of our work would be possible without the right equipment. Notably, automation is absolutely essential for large scale, epidemiological exposure studies. At the CDC, we used Fluid Management Systems (FMS) sample preparation and Thermo Scientific DFS™ Magnetic Sector gas chromatography-high resolution mass spectrometry (GC-HRMS) systems.
But... I’m an analytical scientist, so I always want more! In particular, I want lower detection limits. The levels of many POPs have been declining in the environment and in people and unless this can be matched by a parallel increase in detection sensitivity, increased false positives and negatives will reduce our ability to properly monitor the effects of these pollutants. Increased sensitivity also provides better ‘statistical power’ in epidemiological studies. Achieving equivalent power with fewer study participants saves time and money. Better sensitivity can also pick up completely unexpected information; for example, the large number of non-smokers in the US population with elevated serum cotinine – due to passive exposure – only became evident once analytical sensitivity allowed cotinine measurements at <5 ng/mL.
Down to the attogram level
Clearly, enhanced sensitivity is an ongoing need that demands continuing innovation – especially, to gain the robustness we need. CDC has been working on cryogenic zone compression (CZC) of GC analyte peaks, which when coupled to Magnetic Sector HRMS gives exquisite sensitivity, with very clearly defined peaks at very low sample quantities; for example, ~300 ag for TCDD. And by using GC×GC, we can also benefit from increased peak capacity. We used a GC×GC-TOF MS approach to resolve and quantify 59 analytes found in the human body from one injection, with obvious implications for efficiency and speed in all kinds of applications that demand complex sample analysis.
One of the newest and most exciting approaches is cryogenic zone compression (CZC) GC×GC-HRMS. The first experiments using the CZC approach in combination with Magnetic Sector MS for Dioxins and POPs were realized at the CDC laboratories in Atlanta. In brief, the entire peak of interest is cryotrapped in a single event in one column before being reinjected into the second column (2). The approach produces a very tight, highly-focused peak and gives investigators the option of increasing the sensitivity of the system for particular compounds, as required.
The latest technical advance by Thermo Fisher Scientific developed CZC further into what is now called time-controlled cryogenic zone compression or “t-CZC” (3) see Figure 1. CDC has found that t-CZC gives a clear gain in sensitivity, and furthermore is user-friendly in that you can change between t-CZC and normal operation without needing to change the hardware – even special software is not mandatory. Today, time-controlled CZC is an ongoing development project within Thermo Fisher Scientific, working towards a potential commercially available solution. CDC and other collaborators have been involved for testing and application of this approach on different analytical challenges.
The development of these kinds of techniques – and the extraordinary sensitivity they confer – is allowing us to measure ever lower in terms of detection sensitivity, below the femtogram level and into the attogram range. Soon, we’ll be able to test John Taylor’s hypothesis that “...concentrations of 1 in 1018 of almost any substance can be expected to be present in almost any sample.” We are close to being able to do that now. There’s still work to be done on improving the repeatability and reliability of the new systems and to make them commercially available. But will we get there? Yes, I think we will.
References
- Patterson et al., “Correlation between serum and adipose tissue levels of 2,3,7,8-tetrachlorodibenzo-p-dioxin in 50 persons from Missouri”, Arch Environ Contam Toxicol 17: 139-143 (1988). Patterson et al., “Cryogenic zone compression for the measurement of dioxins in human serum by isotope dilution at the attogram level using modulated gas chromatography coupled to high resolution magnetic sector mass spectrometry”, J. Chromatography A 1218: 3274-3281 (2011). Krumwiede et al., “Time controlled cryogenic zone compression (T-CZC): a novel gas chromatographic tool for increasing sensitivity”, Chromatography Today (Feb/Mar 2012).





