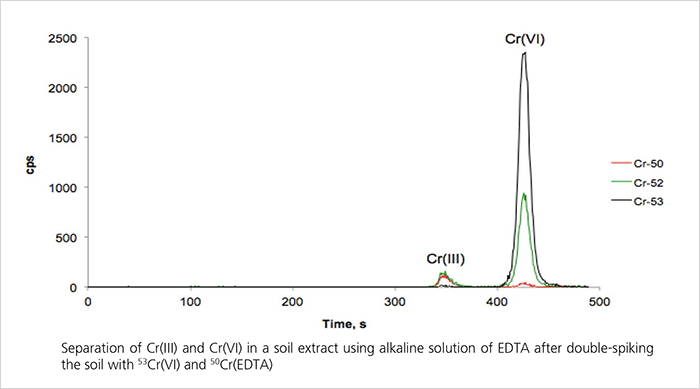
Soil usually contains small amounts of chromium that stem from weathering rocks or anthropogenic sources. The speciation between trivalent chromium – Cr(III) – and hexavalent chromium – Cr(VI) – is important due to the toxic nature of the latter. Here, the two species are separated as Cr(III)-EDTA complex and chromate on a Metrosep A Supp 4 - 250/4.0 column. For detection, speciated isotope dilution mass spectrometry is applied.