I gave a presentation at the 10th International Symposium on Recent Developments in POPs Analysis, held in Prague, Czech Republic, earlier this year (you can see that presentation here: tas.txp.to/0116/Holgado). Why was I there? Essentially, to expand on an investigation that we presented at the 19th International Mass Spectrometry Conference in Kyoto, Japan, in 2012. Back then, we wanted to demonstrate some new ideas of utilizing mass spectrometry for the determination of dioxins in environmental and food samples. In particular, our project looked at the suitability of gas chromatography-tandem mass spectrometry (GC-MS/MS) for the analysis of dioxin-like persistent organic pollutants (dl-POPs). In Kyoto, we compared GC-MS/MS to the reference technique of GC-high-resolution (HR)MS – but the world of mass spectrometry moves very quickly indeed. Here, I share our work with the latest systems.
A short lesson
First, let me quickly go over the key groups of compounds that we are particularly interested in when it comes to POPs analysis: i) Organochlorine pesticides (for example, Aldrin, Chlordane, DDT, Dieldrin, Endrin, Heptachlor, Hexachlorobenzene, Mirex and Toxaphene). Despite many of these compounds being banned, they remain a problem. ii) Polychlorinated biphenyls (PCBs). PCBs have many uses, including dielectric and coolant fluids in electrical apparatus, cutting fluids for machining operations, and in heat transfer fluids. iii) Dioxins. Despite also occurring naturally, dioxins are anthropogenic substances that get into the environment. Why are we so concerned? Well, when these substances enter the food and feed chain, they generally hit the headlines as international scale incidents. There will be few of you who do not recall the scandal of dioxin contamination hitting the Belgian poultry industry back in 1999. And as recently as 2008, Italy had to recall mozzarella cheese products, while Ireland recalled pork products. The silver lining is that such failures in the food chain tend to trigger a positive response; for example, the global Stockholm Convention on POPs, and the EU’s introduction of not only new regulations and limits, but also recommended protocols for sampling and analysis. Reacting to new technology Europe Commission Regulation (EU) No 252/2012 specified that GC-HRMS should be used for confirmatory dioxin analysis in food and feed, while GC-MS/MS was permitted as a screening technique. Jump forward to today and Commission Regulation (EU) No 589/2014 specifies the use of either GC-HRMS or GC-MS/MS for confirmatory dioxin analysis in food and feed, recognizing “technical progress and developments” in GC-MS/MS. Notably, the regulation does state that GC-MS/MS is “an appropriate confirmatory method for checking compliance with the maximum level”, whereas GC-HRMS remains the recommended technique for “determination of low background levels in food monitoring, following of time trends, exposure assessment of the population”. In short, this means Magnetic Sector HRMS has been recognized as delivering superior sensitivity, as required for low-level background studies. Additionally, Magnetic Sector GC-HRMS fulfills all requirements for all types of dioxin applications, and is considered the reference standard for dioxin analysis. The EU regulations for food & feed have specific requirements for GC-MS/MS confirmatory methods. i) You must monitor at least two specific precursor ions, each with one specific corresponding transition product ion, for all labeled and unlabeled analytes.ii) There is a maximum permitted tolerance of relative ion intensities of ±15 percent for selected transition product ions compared with calculated or measured values (the average is taken from calibration standards), applying identical MS/MS conditions, in particular collision energy and gas pressure.
iii) The resolution for each quadrupole is to be set equal to or better than unit mass resolution (unit mass resolution: sufficient resolution to separate two peaks one mass unit apart).
iv) For the limit of quantification (LOQ), the method must demonstrate that it is able to distinguish between the blank and cut-off value. A notification level needs establishing for samples that respond below this level.
v) For polychlorinated dibenzodioxins/polychlorinated dibenzofurans (PCDD/PCDFs), the limits of detection (LOD) should be in the higher femtogram range (10–15 g). For most PCB congeners, it is sufficient to set LOQ in the nanogram range (10–9 g). However, to measure congeners similar to dioxin-like PCBs (in particular non-ortho substituted congeners) the lower limit of the working range must reach the lowest picogram (10–12 g).
In terms of workflow, Magnetic Sector GC-HRMS and GC-MS/MS are similar, following the same preliminary stages of sample preparation, extraction (manual or automated) and cleanup before analysis and finally calculating the toxicity equivalency (TEQ).
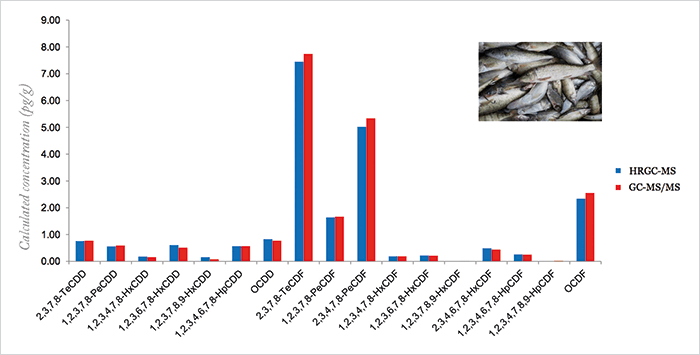
Gold standard
I’ve been working with HRMS - Magnetic Sector analyzer technology for more than 20 years. Currently, we are fortunate enough to enjoy state-of-the-art technology in the form of Thermo Scientific’s DFS™ Magnetic Sector GC-HRMS system, which in my opinion provides gold standard sensitivity for dioxin analysis. Indeed, I think it’s fair to say that it offers excellent performance in terms of sensitivity, robustness, throughput, and flexibility – and that’s more than likely why it is being used by so many dioxin experts around the world. When I started in the lab, Magnetic Sector GC-HRMS seemed out of reach because of the expense and degree of expertise required. I remember I was asked, “Why can’t I analyze dioxins with electron capture detector (ECD)?” – after all, it is very sensitive and relatively cheap technology. I replied, “Go ahead, but you won’t succeed.” Two days later, I was able to see spectra with profiles like the Rocky Mountains, so they moved onto magnetic sector technology... But that hasn’t stopped me searching for alternative solutions. In 2007, we worked on a European Framework project with the University of Barcelona, Spain, using ion-trap MS/MS for analyzing PCDDs and dl-PCBs in food (1). Overall, the data was very good but we had to do a lot of sample clean-up work to get where we needed to be. We felt that it was not a satisfactory technique. Later, we did a collaborative study with The Autonomous University of Nuevo León in Mexico to analyze dioxins in beef samples using a low-resolution MS/MS screening method (a single quadrupole they already had in the lab) (2). It was certainly possible to look for contaminant data, but in the end, we once again concluded that it was not optimal for dioxin analysis.Pushing boundaries, solving problems
Most recently, we have been working with Thermo Fisher Scientific in Manchester, UK, on the development of the TSQ™ 8000 Evo triple quadrupole GC-MS. It is a fast collision cell instrument with proprietary EvoCell technology for high-speed selected reaction monitoring (SRM), precision, and sensitivity. And it features a removable ExtractaBrite™ ion source for high matrix tolerance, as well as easy maintenance. In evaluating its suitability for dioxin analysis, we have investigated various test samples – dried fish, fish, milk powder, feed, fly ash, and sewage sludge – and once again compared it with GC-HRMS. Figure 1 gives one example, and many more are shared in my presentation. How does the instrument measure up to the EU regulations for food & feed? Well, if you look back to the specific requirements for GC-MS/MS confirmatory methods noted earlier, it provides sensitivity that exceeds the rules. So, while Magnetic Sector GC-HRMS is considered as the gold standard in Dioxin analysis with world-wide method compliance and due to its superior sensitivity for lowest level quantitative trace analysis in routine, GC-MSMS has been recognized to be a viable and cost efficient alternative for food & feed analysis in Europe. Moreover one thing is certain; nothing stands still in the world of food analysis and as a proof the most recent innovation to come over the horizon is GC coupled to Orbitrap technology – the Q Exactive™ GC! We’ve already achieved some interesting results and we are excited to see what else the future will bring. GC-Orbi technology will certainly be the new benchmark for powerful unknown compound identification for GC-MS, also in the field of POPs analysis. “If you only have a hammer, every problem is a nail.” Actually, not every problem is the same, is it? So for POPs and dioxins with Magnetic Sector GC-HRMS, GC-MS/MS and now even GC-Orbitrap there is a very complete range of powerful GC-MS technologies available. Each offers specific advantages to the Dioxin or POPs analysis expert, so that a real tailored solution for each specific analytic challenge is possible and can be found. In the end - given all the complex challenges in the world of analysis, you need more than a hammer.References
- J Malavia et al., “Ion-trap tandem mass spectrometry for the analysis of polychlorinated dibenzo-p-dioxins, dibenzofurans, and dioxin-like polychlorinated biphenyls in food”, J Agric Food Chem, 26, 55(26),10531–10539 (2007). PMID: 18052095. L Naccha et al., “Dioxins in beef samples from Mexico using a low resolution GC/MS screening method”, Food Addit Contam Part B Surveill, 3(1), 64–72 (2010). PMID: 24785318.





