In The Story of Our App Note, scientists share the inspiration, challenges, and breakthroughs behind their application research. This edition explores how a newly developed LC-MS/MS method for the anti-HER2 antibody-drug conjugate trastuzumab emtansine could transform pharmacokinetic studies of ADCs – offering unprecedented sensitivity from minimal sample volumes. Scroll down to read the interview with Lakshmanan Deenadayalan, Application Support Scientist, SCIEX, and download the full app note.
What inspired this application?
Drug discovery is evolving with the growing use of antibody drug conjugates (ADCs) for the search for cancer treatments. ADCs are unique in their ability to target cancer cells, delivering a powerful payload directly to tumor cells with incredible specificity. Over 100 ADC-based drugs have been introduced into the market, but there is still no accurate workflow to study their pharmacokinetic (PK) effects in human serum. As such, we set out to develop a robust, sensitive, and reproducible workflow for trastuzumab emtansine (TEx), an anti-HER2 ADC used to treat breast cancer, using the SCIEX 7500+ system.
What was the biggest challenge you faced?
Because there was little available information on the quantitation of trastuzumab emtansine in human serum, we needed to overcome several challenges to develop a robust LC-MS/MS method. The first was selecting the proper mag beads to extract the ADC from the highly complex matrix. Next, the on-bead digestion process needed to be optimized to achieve maximum recovery with minimal processing time. The final challenge was refining the chromatography and MRM method parameters to properly separate the co-eluting peaks. Ultimately, we established a highly robust method for the continuous analysis of ADC samples.
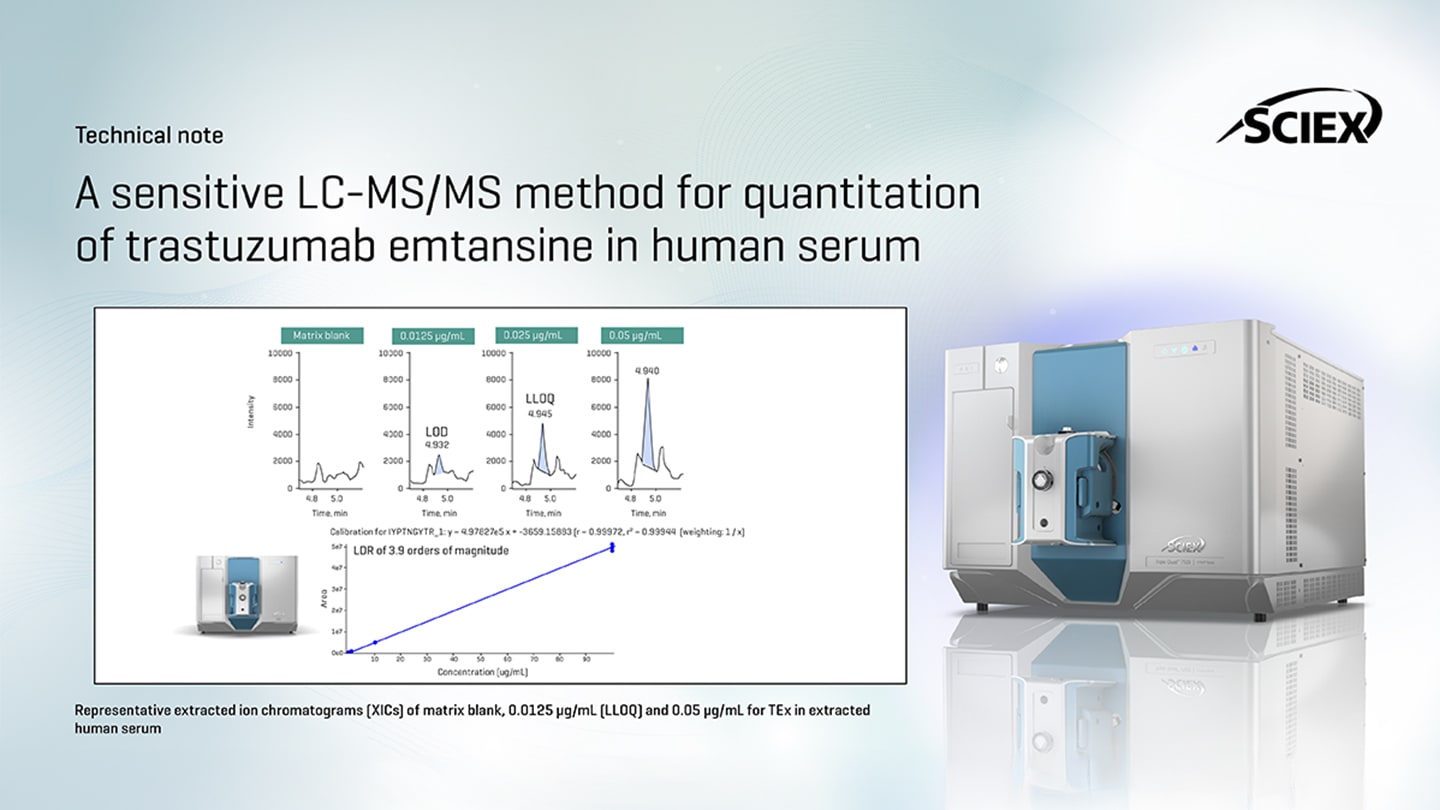
What enabled you to achieve such low detection and quantitation limits?
A combination of several important factors helped us to achieve such low levels of quantitation – 0.025 pg/mL LLOQ and 0.0125 ug/mL LOD. Immunoaffinity capture with Protein A mag beads was a critical step, where the targeted analyte was enriched from the serum. This was followed by an on-bead tryptic digestion to achieve reduced or minimal interference. We also optimized chromatography and MRM method parameters on the highly sensitive SCIEX 7500+ system, demonstrating the capability to build a robust LC-MS/MS method that will reproducibly quantify at low levels with very minimal starting sample volume.
How could this method impact future studies of antibody-drug conjugates?
This LC-MS/MS method could significantly impact future studies on antibody-drug conjugate analysis by providing a reliable analytical workflow – enabling sensitive, accurate, and reproducible quantitation in biological matrices. And with 21 CFR Part-11 compliance, SCIEX OS software supports improved method transfer between laboratories and compliance with regulatory requirements, ultimately accelerating ADC drug development and clinical translation.




