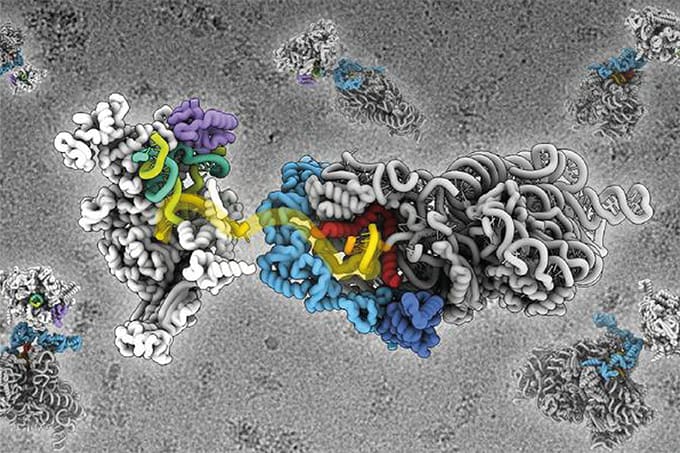
You may be surprised to hear that cryo-EM has been around since the 1950s given its recent, much publicized coming of age. Following the “resolution revolution,” the number of structures resolved by cryo-EM has grown exponentially, reaching close to 3000 in 2019 alone (1). Thanks to cryo-EM, we can now access high resolution structures for integral membrane proteins, including ion channels and GPCRs (G-protein coupled receptors), in their near native state. We can also study epitopes and protein-ligand binding in unprecedented detail; and we have already used cryo-EM to solve structures that had long proven elusive, including the insulin receptor (2). And, of course, it was cryo-EM that revealed the famous spike structure of SARS-CoV-2, paving the way to the development of effective vaccines.
So, the excitement is justified. Going forward, cryo-EM will accelerate the application of “structure based drug design;” specifically, it will enable the development of structural and mechanistic understanding that has previously been inaccessible, helping us map routes to success rather than approaching drug development with empiricism alone as our guide.
And yet, I’d like to temper the prevailing enthusiasm with a few words of caution. Cryo-EM is complicated and difficult. Instrumentation is expensive: a high-resolution set-up costs around US$10 million. Measurements call for a daunting level of computational infrastructure and expert analytical input, which remains in relatively short supply. Last but certainly not least, cryo-EM is not suitable for all molecules and sample quality is critical. I’d like to dig a little deeper into this last point as failures are often directly attributable to sample quality and inadequate preparation.
Interestingly, the technological improvement responsible for the resolution revolution is partially to blame. The defining advance in detector technology was to enable the capturing of a short video (within a few seconds) during data collection – multiple frames rather than single images. But bombarding a frozen sample with electrons induces specimen motion; and for single images, this means lack of clarity – while a movie allows frame alignment, delivering high resolution.
Potentially problematic samples can sometimes be detected via techniques such as dynamic light scattering (DLS) and negative staining. DLS quantifies size rather than molecular mass, so if results suggest a large molecule with a small mass, then a problematic elongated structure is more likely. Negative staining will provide an initial visualization of the aggregation state and monodispersity of the sample. However, obtaining a sample that will deliver valuable results can be the most demanding step in a cryo-EM analysis.
With appropriate sample preparation? Well, cryo-EM offers the ability to see multiple conformations and domains – the closed and open configuration of an enzyme working with a ligand, for example. And a primary advantage of cryo-EM is its ability to work with large biomolecular complexes, which are intrinsically flexible molecules that are unsuitable for crystallography (they cannot be crystallized).
I’m a crystallographer by background, and when I first began to learn about cryo-EM I was privileged to spend two years working alongside experts at the New York Structural Biology Center. There’s really no substitute for working with other experts when it comes to cryo-EM; it gave me the foundation I needed to apply cryo-EM in a productive way in my own lab. Cryo-EM is unquestionably a breakthrough technique and the knowledge accumulated in the last three years alone is staggering.
Going forward it will help us to drive drug discovery to new levels of efficiency, but the old techniques still have value. We don’t need cryo-EM for every molecule or to always work to the highest levels of resolution. Applying cryo-EM judiciously and effectively is the key to unleashing its full potential.
References
- Nature, “Revolutionary cryo-EM is taking over structural biology” (2020). Available at: https://go.nature.com/3wKobcO
- G Scapin et al. “Structure of the insulin receptor-insulin complex by single-particle cryo-EM analysis,” Nature, 556, 7699, 122 – 125 (2018). PMID: 29512653.