Today’s laboratories have to deal with an increased number of samples; throughput is a progressively more important factor. Solutions have been presented, for example, highly efficient narrow bore columns. However, operating 0.10 mm ID columns demands specific instrumentation and skills. Moreover, use is limited by sampling, loadability, durability, and the measurement of “fast” peaks. The use of 0.15/0.18 mm ID capillaries has been better accepted.

Selectivity is one of the “forgotten” factors affecting separation in gas chromatography. In the early days, when packed columns were the standard, many types of stationary phases were used to optimize separation because only 5000-10,000 theoretical plates were available. A selective stationary phase simply had to be chosen. With the introduction of the capillary column, selectivity became less important: by using 30-meter columns, over 120,000 theoretical plates are easily generated. Ditching selectivity for plate number translated into generalized phase recommendations, with only a polar (polyethylene glycol) and a non-polar (polydimethyl siloxane) type capillaries covering the majority of separations. Even today, the majority of separations can be performed with only two phases (polar and non-polar), but, crucially, if faster analysis is also required, it is essential to approach the challenge in the right way.
One of the best ways to reduce analysis time is to use the selectivity of the stationary phase (see “Back to Basics”).
Here is the (simplified) resolution equation:
Here is the (simplified) resolution equation:
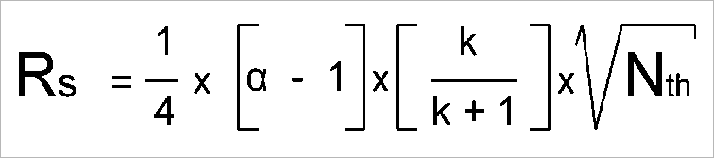
Resolution (R) between two compounds depends on plate count (Nth), retention factor (k), and the selectivity factor (α), but their relative impacts must be put in perspective.
Increasing k
Increasing k will only be of interest if k is very low as R increases and then near linear with increase of k. At higher k (longer retention time), the k-term becomes “1”, meaning no additional contribution to R. The k of an existing column can be changed with temperature. k can also be changed using a column with a different phase-ratio.
Increasing Nth
Increasing plate count will always impact R, but the contribution is not large (square root value); a 4x longer column is needed to get 2x increase in resolution. Additionally, retention times are increased, making analysis time longer, and peaks broader and therefore lower. Longer columns also have a higher cost.
Increasing α
Increasing the selectivity factor will increase resolution linearly. The best way to optimize any separation is to choose the most selective stationary phase.
As the selectivity factor has the biggest impact, a small change translates into a big difference in the separation of target analytes. And by using highly selective materials, shorter columns can be used, resulting in faster separations. For the fastest analyses, one can use the best of both worlds: selectivity and small diameter columns. Furthermore, selectivity can be manipulated by temperature (see “Manipulation of selectivity”).
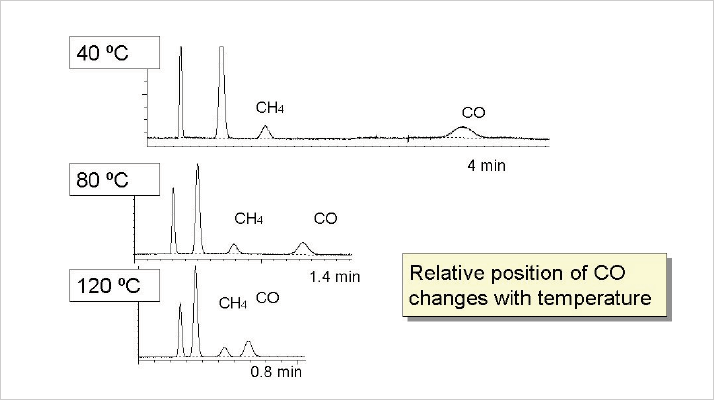
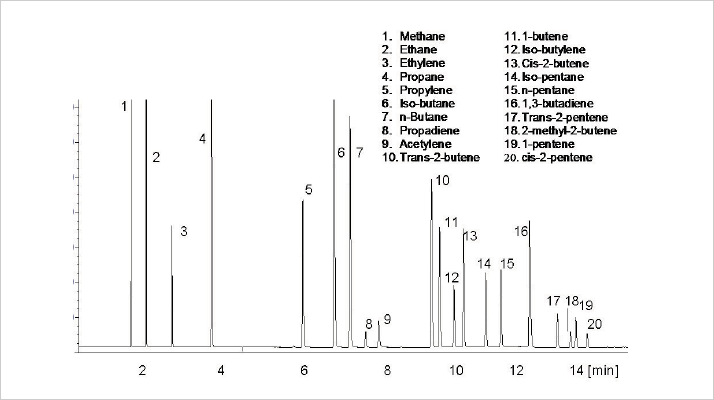
Although selectivity of a stationary phase is a “fixed” parameter, it is also a function of the temperature. Changing operating temperature will affect the relative position of components in chromatograms; sometimes separations improve, sometimes they get worse. For adsorbents, the stationary phase acts more “polar” when the elution temperature of components is reduced, which means that polar components elute later at lower temperatures. This effect is of particular interest with alumina columns, where polar hydrocarbons like acetylene and propadiene can be positioned before or after the n-butane peak, just by changing temperature conditions. With a 5A molecular sieve the effect is very strong (see Figure 5). The lower the temperature, the more polar the column behaves, resulting in much higher retention for carbon monoxide.
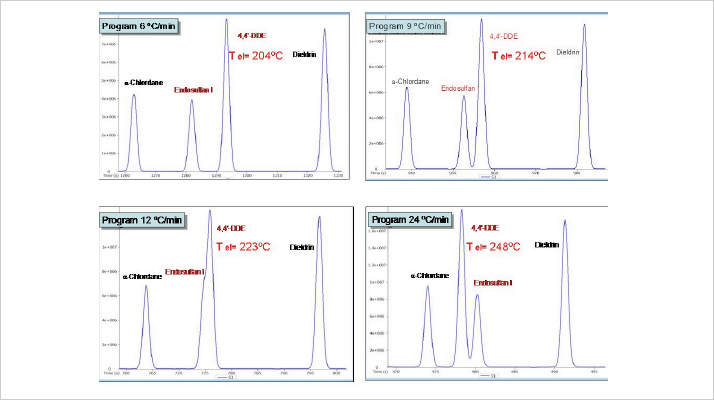
For liquid stationary phases, reduced temperature results in a reduced “polar” nature. For such phases it would be desirable to use thicker films to enhance retention and run columns at a higher temperature to make best use of the higher polarity. The challenge is that polar phases also suffer from decreased temperatures stability. Fortunately, the use of silphenylene and trifluoropropyl type building blocks allows phases to exceed 340 °C with an acceptable base line. Figure 6 shows an example of temperature impact using a phase that was developed for optimal selectivity in the separation of chlorinated pesticides under certain conditions. By changing the oven or flow conditions, different selectivity effects were observed; when faster temperature programming was used at constant flow, pesticides eluted at higher temperatures. The same effects can be obtained when the flow is changed using a fixed temperature program. This selectivity effect usually increases with the polarity of the stationary phase, so it is always important (and interesting) to run a separation at different temperature programs, to learn how and which peaks “move”.
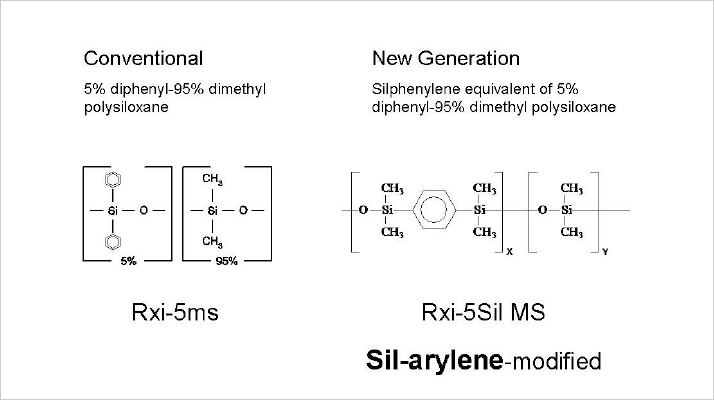
It was the first thing that my teacher taught me about selectivity: the most selective phase is the one that looks most like the components you are analyzing. Initially, this appears a little strange; interactions will be similar for all component types, so how can I achieve separation? While true as an initial, general rule – separation is achieved by the size (molecular weight) of the component – in reality, the goal is to achieve a difference in retention based on molecular interactive forces that determine solubility. Such interactions are based on dispersion, formation of hydrogen bridges, van der Waals forces, dipoles, and steric hindrance. The best way to change the forces above is by using functional groups in the stationary phase that show maximal interaction with the analytes. Alcohols separate the best on a polyethylene glycol type phase and hydrocarbons will do well on non-polar phases like squalane or polydimethyl siloxane. Over the last decade, we’ve made big breakthroughs in stabilizing stationary phases using silphenylene groups. We found that it was possible to reduce the breakdown reaction of diphenyl siloxanes by building the phenyl group in the main chain (see Figure 1) (1). Such phases show lower bleed, but also show different selectivity – something that is not always made clear by vendors – which can lead to strange examples of peak elution shifts; that’s why some vendors supply traditional 5% diphenyl siloxane alongside the stabilized silphenylene phase.
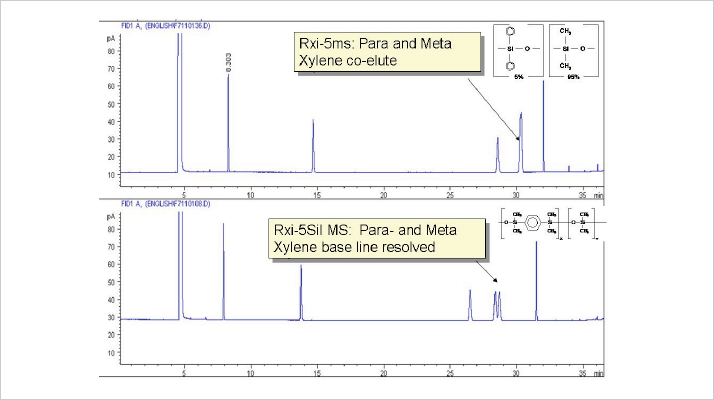
For certain applications the silphenylene-stabilized phase offers better separation. Figure 2 shows the separation of aromatics on both phases under similar conditions and dimensions. The silphenylene-stabilized phase shows a separation of para- and meta xylene, while the 5% phenyl phase is unable to resolve the peaks. This is interesting as the separation was only possible using polar PEG and TCEP type phases. With more complex samples, there will be greater differences; we have seen issues with many compound classes (2-4). Modification of the backbone can do a lot for specific separations and several unique phase chemistries have been developed based on this premise for dioxins, chlorinated pesticides and PCBs, for example.
Light hydrocarbons
For the separation of light hydrocarbons, a phase with high retention and selectivity is required. The most selective phase for hydrocarbons is squalane, but it cannot be deposited in sufficiently thick films to generate enough retention. The second choice is 100% polydimethyl siloxane – the methyl groups ensure good interaction with hydrocarbons and film thickness of 5 µ can be easily achieved on 0.32mm ID capillaries. However, if 100% dimethyl siloxane is unable to resolve unsaturated hydrocarbons (as is the case in Figure 3), more selectivity is required and adsorption chromatography becomes an attractive option. Alumina interacts with hydrocarbons in a unique fashion, which allows complete separation of C1-C5 hydrocarbons at temperatures above 60 °C (see Figure 4). Furthermore, the high selectivity means that the analysis time can be reduced to a minimum using high linear gas velocities and high-speed programming.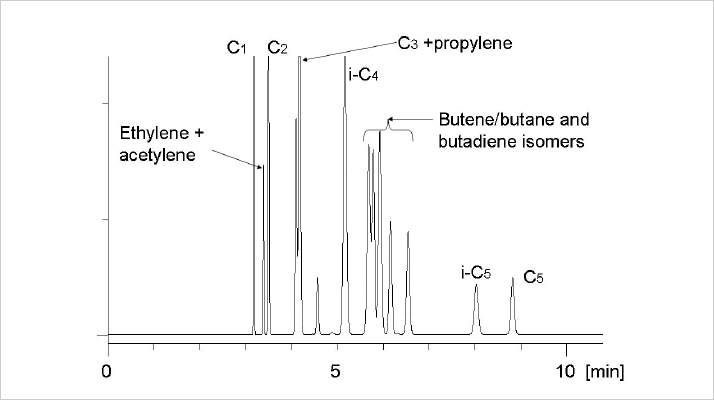
Polycyclic aromatic hydrocarbons (PAH)
PAHs must be analyzed at low levels and elute at higher temperatures, so a low bleed system is required. The Silphenylene technology as previously discussed provides very stable solutions. Figure 7 shows the baseline resolution of all 16 traditional EPA 610 PAHs using a Silphenylene stabilized phase on a 30m x 0.25mm capillary. Benzo(g,h,i)perylene elute at 310 °C on a straight baseline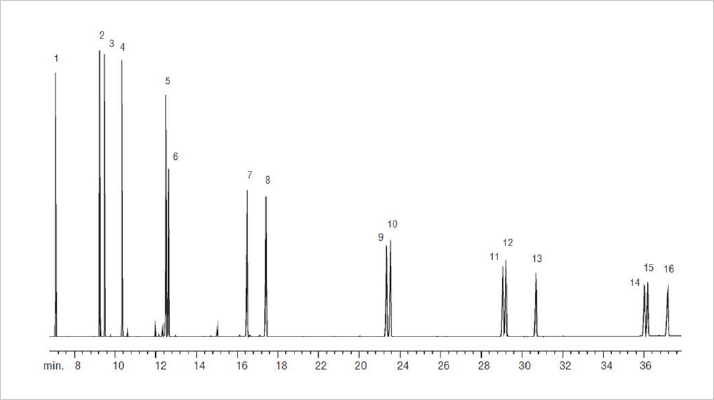
However, the last ten years have seen more demand for measuring an increased number of PAHs, particularly in food analysis. In fact, the new list of current interest to the European Food Safety Agency is quite challenging – several isobaric PAHs must be quantified, which is not only a problem for flame ionization detection but also for mass spectrometry. Chromatographic separation is essential. Specific examples are the separation of benzo-fluoranthene isomers and the separation of chrysene-triphenylene. Increasing the Silphenylene content (for example, as in Rxi-17Sil MS, see Figure 8) increases selectivity for PAHs and provides the highest possible signal-to-noise ratio for trace monitoring; however, it does not offer enough selectivity for the separation of triphenylene-chrysene. Separation of triphenylene-chrysene can be obtained on a Rxi-XLB phase, but only at the expense of fluoranthene separation (5).
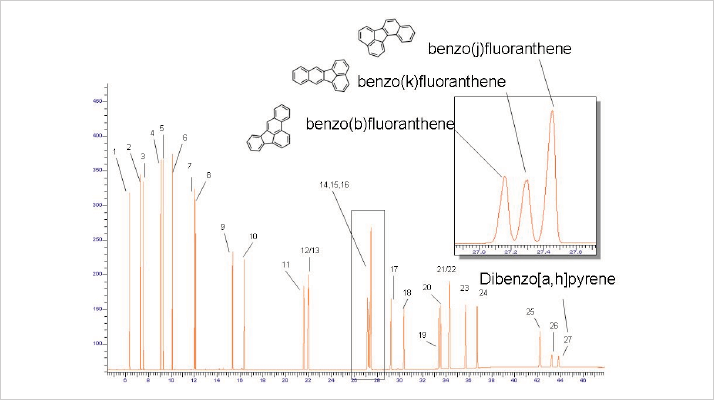
A new selective phase for PAH
At Restek, we recently developed a new high temperature phase that incorporated groups that specifically interact with both target PAH structures, which separates all relevant PAHs (see Figure 9), is stable up to 360 °C, and was found to be robust for repeated injections of PAH extracts from food. Dibenzo pyrene elutes at 350 °C; note the stable baseline at this very high temperature.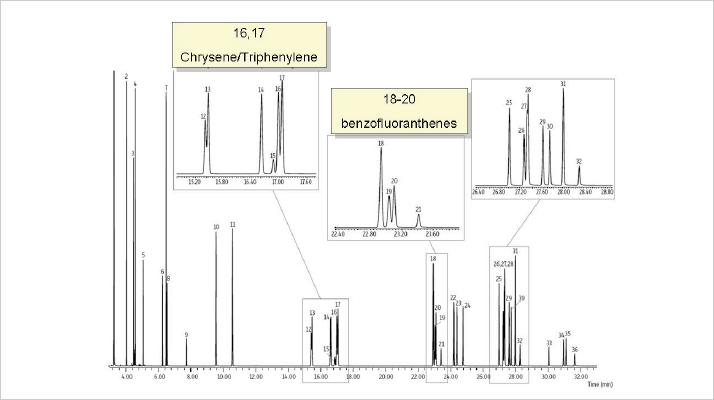
Smart use of stationary phase selectivity allows users to achieve the most efficient separations in the shortest possible time. As such, it should be the first port of call in challenging separations, well ahead of considering column dimensions and operational conditions. In practice, and especially in method development, one has to think about the structures of the compounds of interest and relate that information to the stationary phases available. This is valid for both GC and LC separations (6). The leading commercial companies offer a selection of GC phases that have already been optimized for a certain applications – such phases can be easily recognized as the name is often linked with the application. If there are no application-specific phases, one can use online chromatogram databases and there are even specific programs that can “model” separations.
Acknowledgements
Special thanks to Amanda Rigdon, Jack Cochran, Roy Lautamo, Shawn Reese, Jason Dunbar, Barry Burger, and Chris English for providing chromatograms and stimulating discussions.
References
- J. de Zeeuw et al., “Development of a newplatform of highly inert fused-silica capillary columns,” American Lab., 40 (12), 11-15 (2008). blog.restek.com/?p=617 blog.restek.com/?p=7090 blog.restek.com/?p=619 bit.ly/12t2Nj8 bit.ly/154RxN2




